ABOUT
EXPERIENCE
With over 12 years of experience, GMPharma ensures that Validation and Project Management services meet the highest industry standards, providing tailored solutions to support your success.
MISSION
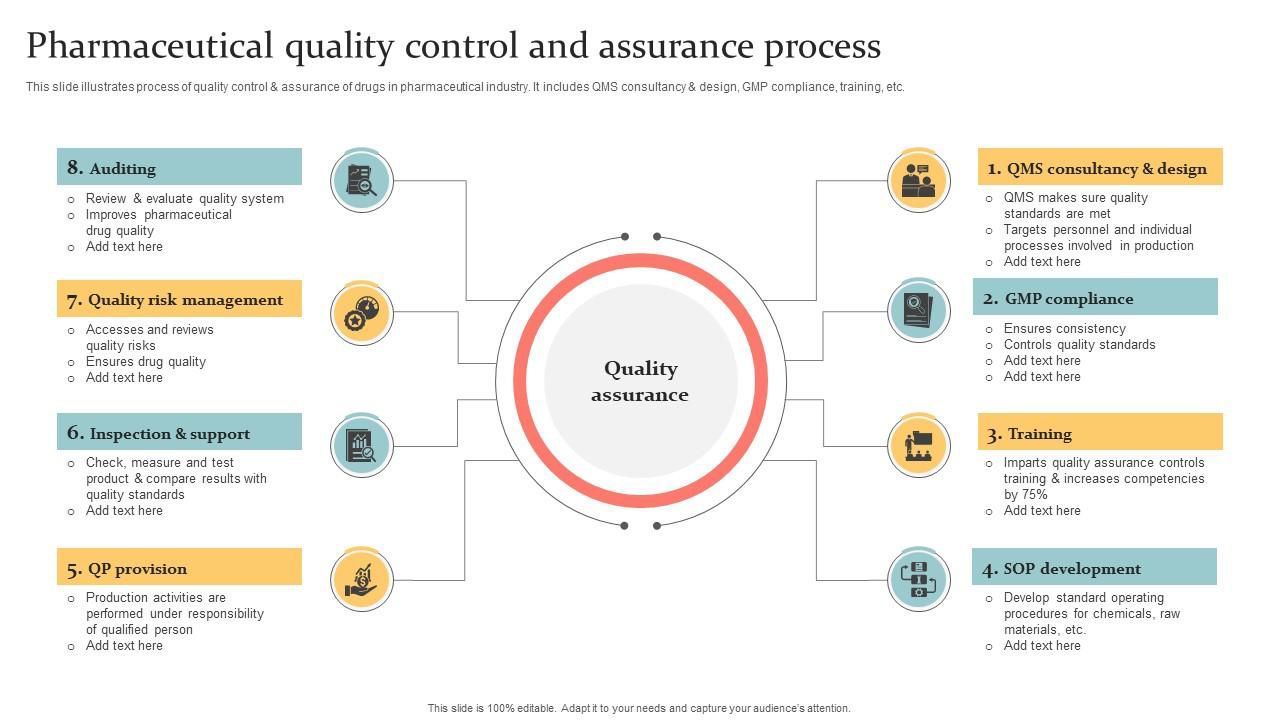
I specialise in effectively managing projects, adhering to all regulatory requirements, and optimising the Quality Management System (QMS), I am dedicated to delivering the highest quality outcomes for my clients:
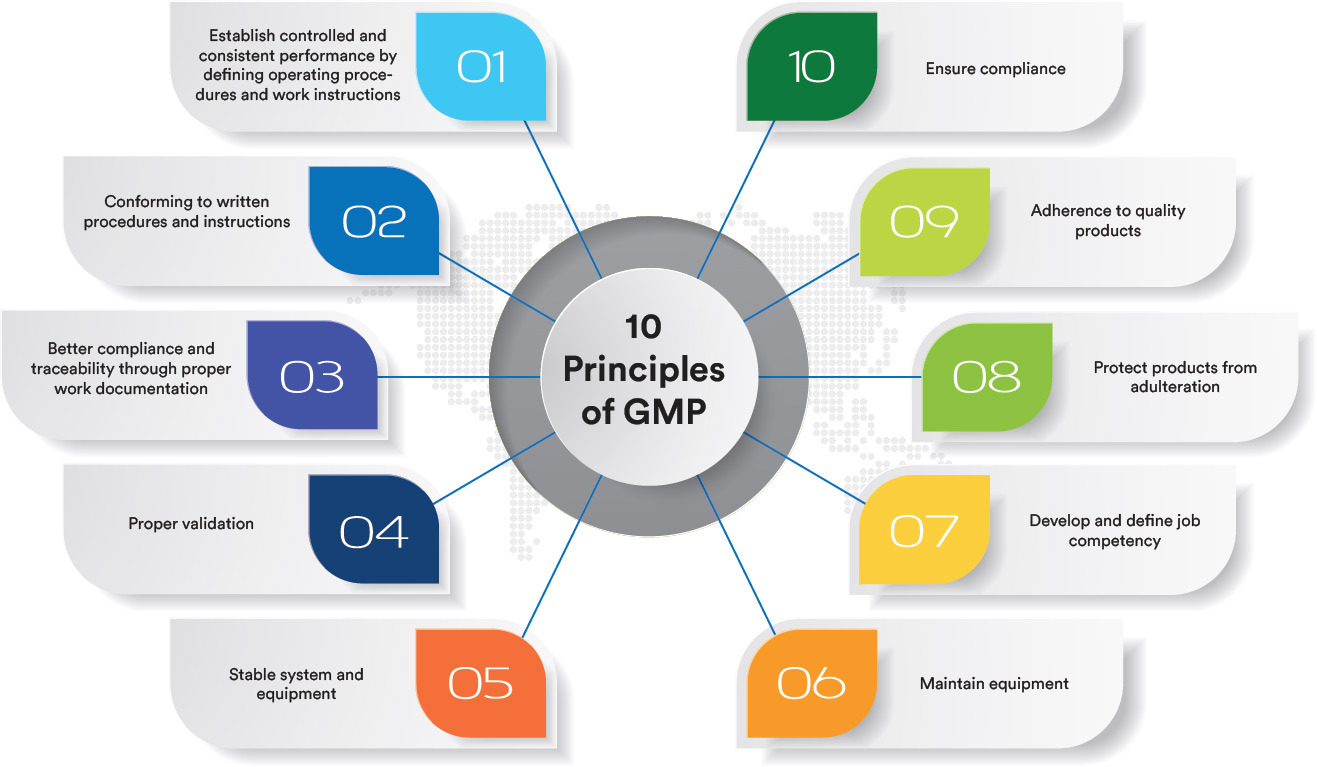
• Validation & Qualification (Facilities, Equipment, Systems)• Quality Assurance & GMP Compliance (EMA/FDA/TGA)
• GxP Facility Design & Capital Project Management
• Continuous Improvement & Inspection Preparation
SERVICES
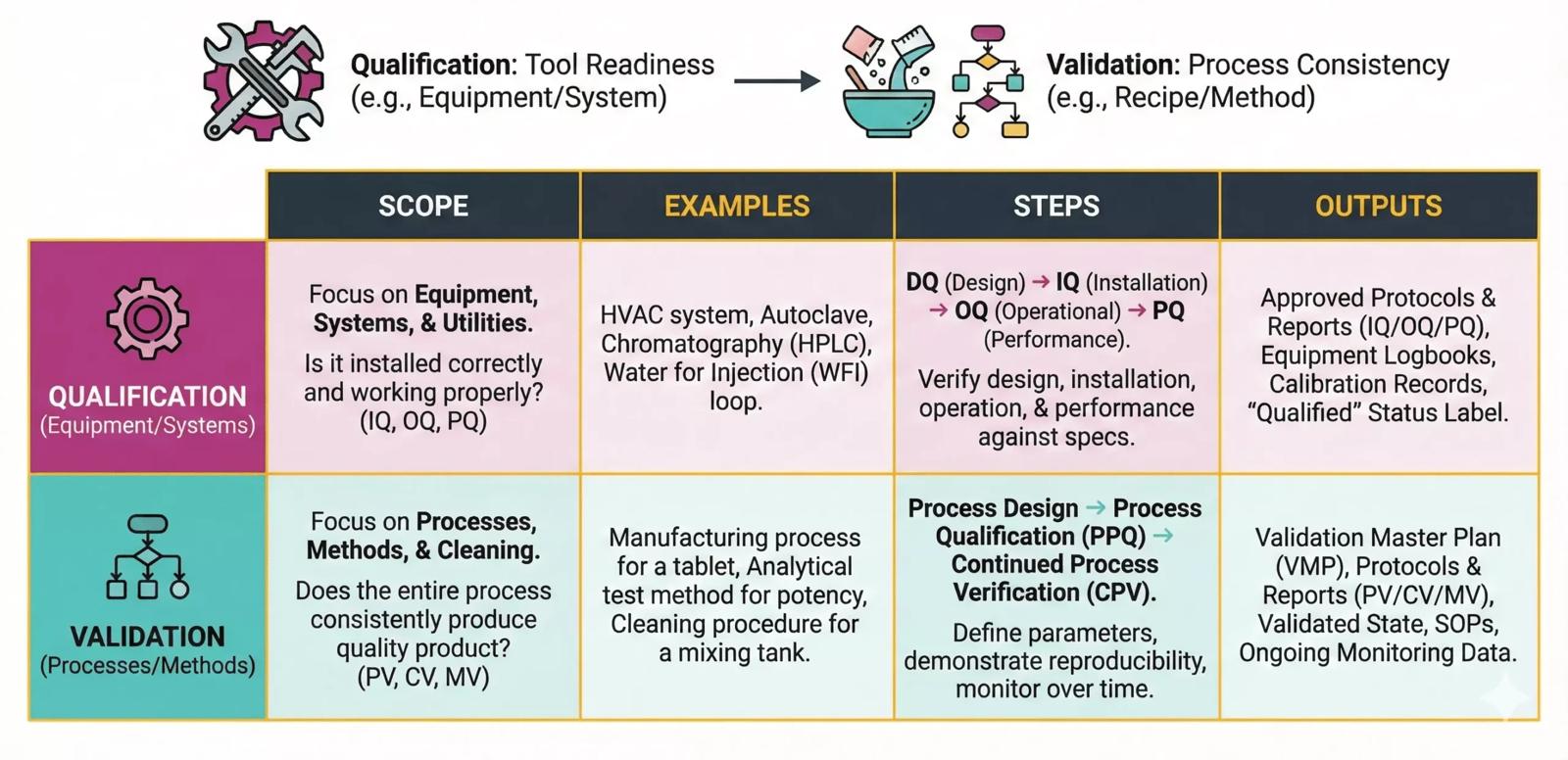
At GMPharma, I’m your go-to expert for Qualification and Validation. I use the latest risk-based Computer System Validation (CSV) methods to make sure your systems are ready for inspections by the TGA, FDA, EMA, MHRA, and other regulators. With a solid scale-up and development background across the pharmaceutical, biotech, medical device, clinical research, and software industries, I’m here to help you stay on track with compliance.
GMP Readiness and Audits/Certification
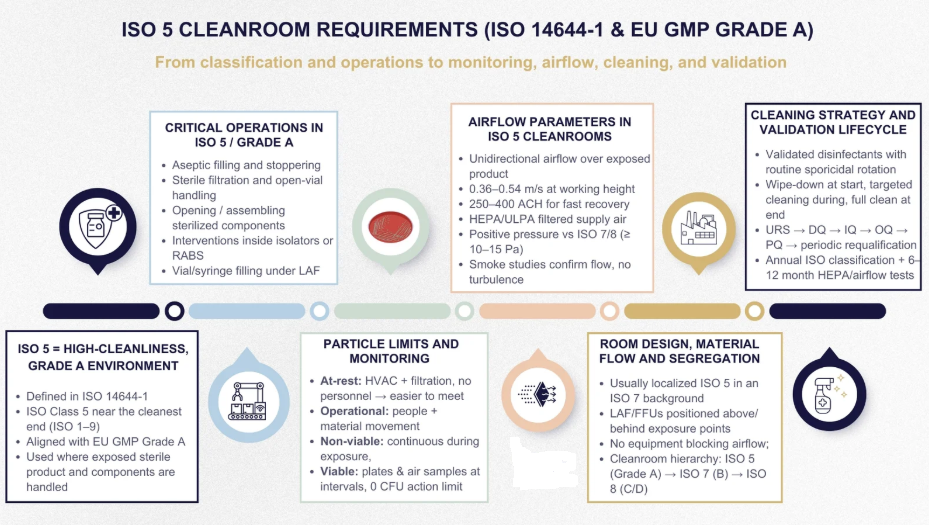
I offer full-on audit services in various GxP areas worldwide, including gap analysis and risk assessment, using my team’s diverse language skills to save you time and money. My auditors are well-versed across the entire development and regulatory landscape, covering GMP readiness in compliance with TGA, FDA, MHRA, PIC/S, EU, ISO13485, ISO14644 and ISO9001.Quality Management Systems (QMS/eQMS)
I work with your team to provide hands-on support for QMS software and tools (Change Controls, CAPA, NCR, etc) within your quality system requirements and standards With years of experience in consulting and operational roles, I help you navigate the process and defend your CAPA systems to regulators while optimising your program.
Other Services
- Validation Consultancy
- Vendor Assurance/Supplier audits systems
- Facility/Cleanroom Design and Setup
- Audit Findings and Responses
- Technical Writing


Recent Updates
- Validation & Qualification Update | Australia – Moving Beyond Template-Based Compliance
- GMP & TGA Update | Increased Focus on Data Integrity and System Validation
- Why Adopting TGA Makes Freeze Dryer Qualification a Breeze
- ⚙️ Equipment Maintenance: The Unsung Hero of Your Workday
- How to Qualify Cleanrooms in Australia (Without Falling Asleep Doing It)
- The Future of GMP Compliance: Are You Ready for 2026?
- Validation in 2026: It’s No Longer a Checkbox — It’s Your Competitive Edge
- Is Your CAPA System Audit-Ready in 2026? Most Aren’tIs Your CAPA System Audit-Ready in 2026? Most Aren’t
- 🇦🇺 How Is GMP Manufacturing Done in Australia — and How Does It Compare to the US and EU?
WHY GMPharma?
GMPharma leverages cutting-edge tools and technologies to stay at the forefront of the pharmaceutical and biotechnology industries. With a commitment to innovation, investment in advanced systems, and continuous expansion of a global network, GMPharma provides top-tier quality services while reducing environmental impact. Focused on excellence in every aspect of GMP compliance, GMPharma goes beyond expectations to deliver what truly matters to clients. Quality excellence is in GMPharma’s DNA.